Cam Plants Keep Stomata Closed in Daytime Thus Reducing Loss of Water. They Can Do This Because They

Crassulacean acrid metabolism, also known every bit CAM photosynthesis, is a carbon fixation pathway that evolved in some plants equally an adaptation to arid conditions[ane] that allows a institute to photosynthesize during the day, but only exchange gases at night. In a establish using total CAM, the stomata in the leaves remain shut during the day to reduce evapotranspiration, but they open at dark to collect carbon dioxide (COii) and allow it to diffuse into the mesophyll cells. The CO2 is stored as the four-carbon acid malic acid in vacuoles at night, and then in the daytime, the malate is transported to chloroplasts where information technology is converted back to COtwo, which is so used during photosynthesis. The pre-collected CO2 is full-bodied around the enzyme RuBisCO, increasing photosynthetic efficiency. This mechanism of acid metabolism was first discovered in plants of the family unit Crassulaceae.
Historical background [edit]
Observations relating to CAM were outset fabricated by de Saussure in 1804 in his Recherches Chimiques sur la Végétation.[2] Benjamin Heyne in 1812 noted that Bryophyllum leaves in India were acidic in the morn and tasteless past afternoon.[iii] These observations were studied further and refined by Aubert, E. in 1892 in his Recherches physiologiques sur les plantes grasses and expounded upon past Richards, H. M. 1915 in Acidity and Gas Interchange in Cacti, Carnegie Establishment. The term CAM may take been coined by Ranson and Thomas in 1940, but they were non the first to discover this cycle. It was observed by the botanists Ranson and Thomas, in the delicious family Crassulaceae (which includes jade plants and Sedum).[4] Its proper noun refers to acrid metabolism in Crassulaceae, non the metabolism of "crassulacean acrid", a nonexistent chemical entity.
Overview: a two-part cycle [edit]

CAM is an accommodation for increased efficiency in the use of h2o, and then is typically found in plants growing in barren conditions.[5] (CAM is establish in over 99% of the known 1700 species of Cactaceae and in almost all of the cactii producing edible fruits.)[half dozen]
During the night [edit]
During the night, a plant employing CAM has its stomata open, allowing COii to enter and exist stock-still every bit organic acids by a PEP reaction similar to the C4 pathway. The resulting organic acids are stored in vacuoles for later use, as the Calvin bicycle cannot operate without ATP and NADPH, products of calorie-free-dependent reactions that practise not take place at night. [7]

Overnight graph of CO2 absorbed past a CAM plant
During the twenty-four hours [edit]
During the day the stomata close to conserve water, and the CO2-storing organic acids are released from the vacuoles of the mesophyll cells. An enzyme in the stroma of chloroplasts releases the COii, which enters into the Calvin bicycle so that photosynthesis may have place.[ citation needed ]
Benefits [edit]
The nigh important benefit of CAM to the plant is the ability to leave most leaf stomata closed during the solar day.[viii] Plants employing CAM are almost common in arid environments, where water comes at a premium. Being able to go on stomata closed during the hottest and driest role of the twenty-four hour period reduces the loss of water through evapotranspiration, allowing such plants to grow in environments that would otherwise be far too dry out. Plants using only C3 carbon fixation, for example, lose 97% of the h2o they accept up through the roots to transpiration - a loftier cost avoided by plants able to utilize CAM.[nine] [ What percentage is lost in CAM plants? ]
Comparing with C4 metabolism [edit]

The C4 pathway bears resemblance to CAM; both act to concentrate CO2 effectually RuBisCO, thereby increasing its efficiency. CAM concentrates information technology temporally, providing CO2 during the day, and not at night, when respiration is the ascendant reaction. C4 plants, in dissimilarity, concentrate CO2 spatially, with a RuBisCO reaction center in a "bundle sheath jail cell" being inundated with CO2. Due to the inactivity required by the CAM mechanism, C4 carbon fixation has a greater efficiency in terms of PGA synthesis.
There are some Cfour/CAM intermediate species, such as Peperomia camptotricha, Portulaca oleracea, and Portulaca grandiflora. The two pathways of photosynthesis can occur in the same leaves only not in the same cells, and the two pathways cannot couple and can only occur side by side[10]
Biochemistry [edit]

Plants with CAM must control storage of CO2 and its reduction to branched carbohydrates in space and time.
At depression temperatures (oft at night), plants using CAM open their stomata, COii molecules lengthened into the spongy mesophyll'south intracellular spaces and then into the cytoplasm. Here, they tin meet phosphoenolpyruvate (PEP), which is a phosphorylated triose. During this time, the plants are synthesizing a protein called PEP carboxylase kinase (PEP-C kinase), whose expression tin can be inhibited by high temperatures (frequently at daylight) and the presence of malate. PEP-C kinase phosphorylates its target enzyme PEP carboxylase (PEP-C). Phosphorylation dramatically enhances the enzyme's capability to catalyze the germination of oxaloacetate, which tin be subsequently transformed into malate by NAD+ malate dehydrogenase. Malate is then transported via malate shuttles into the vacuole, where it is converted into the storage course malic acid. In contrast to PEP-C kinase, PEP-C is synthesized all the time but nearly inhibited at daylight either by dephosphorylation via PEP-C phosphatase or directly by binding malate. The latter is non possible at low temperatures, since malate is efficiently transported into the vacuole, whereas PEP-C kinase readily inverts dephosphorylation.
In daylight, plants using CAM close their baby-sit cells and discharge malate that is subsequently transported into chloroplasts. There, depending on plant species, it is cleaved into pyruvate and CO2 either by malic enzyme or past PEP carboxykinase. CO2 is so introduced into the Calvin bicycle, a coupled and self-recovering enzyme organisation, which is used to build branched carbohydrates. The past-product pyruvate can be further degraded in the mitochondrial citric acid cycle, thereby providing additional CO2 molecules for the Calvin Cycle. Pyruvate tin also be used to recover PEP via pyruvate phosphate dikinase, a high-energy step, which requires ATP and an additional phosphate. During the post-obit cool dark, PEP is finally exported into the cytoplasm, where it is involved in fixing carbon dioxide via malate.
Use by plants [edit]
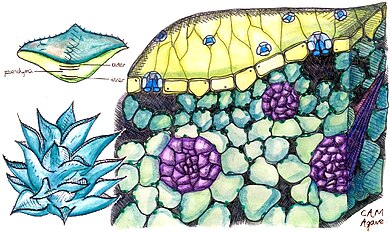
Cross section of a CAM (Crassulacean acid metabolism) plant, specifically of an agave leaf. Vascular bundles shown. Drawing based on microscopic images courtesy of Cambridge University Plant Sciences Section.
Plants utilise CAM to different degrees. Some are "obligate CAM plants", i.e. they use just CAM in photosynthesis, although they vary in the amount of CO2 they are able to store as organic acids; they are sometimes divided into "strong CAM" and "weak CAM" plants on this footing. Other plants testify "inducible CAM", in which they are able to switch between using either the C3 or C4 machinery and CAM depending on environmental atmospheric condition. Another grouping of plants apply "CAM-cycling", in which their stomata do non open up at dark; the plants instead recycle CO2 produced by respiration likewise every bit storing some COtwo during the 24-hour interval.[five]
Plants showing inducible CAM and CAM-cycling are typically found in conditions where periods of water shortage alternating with periods when water is freely available. Periodic drought – a feature of semi-arid regions – is one cause of water shortage. Plants which abound on copse or rocks (as epiphytes or lithophytes) also experience variations in water availability. Salinity, high light levels and food availability are other factors which take been shown to induce CAM.[5]
Since CAM is an adaptation to arid conditions, plants using CAM often display other xerophytic characters, such as thick, reduced leaves with a low surface-area-to-book ratio; thick cuticle; and stomata sunken into pits. Some shed their leaves during the dry out season; others (the succulents[11]) store water in vacuoles. CAM too causes taste differences: plants may have an increasingly sour sense of taste during the night yet become sweeter-tasting during the solar day. This is due to malic acid being stored in the vacuoles of the plants' cells during the night then being used up during the day.[12]
Aquatic CAM [edit]
CAM photosynthesis is also found in aquatic species in at least 4 genera, including: Isoetes, Crassula, Littorella, Sagittaria, and peradventure Vallisneria,[13] being found in a variety of species e.g. Isoetes howellii, Crassula aquatica.
These plants follow the same nocturnal acid accumulation and daytime deacidification as terrestrial CAM species.[14] Notwithstanding, the reason for CAM in aquatic plants is not due to a lack of bachelor water, but a limited supply of COii.[13] CO2 is limited due to slow diffusion in water, 10000x slower than in air. The problem is specially acute under acid pH, where the only inorganic carbon species present is COii, with no available bicarbonate or carbonate supply.
Aquatic CAM plants capture carbon at nighttime when it is abundant due to a lack of competition from other photosynthetic organisms.[14] This also results in lowered photorespiration due to less photosynthetically generated oxygen.
Aquatic CAM is nigh marked in the summer months when there is increased competition for COii, compared to the winter months. All the same, in the winter months CAM still has a significant role.[15]
Ecological and taxonomic distribution of CAM-using plants [edit]
The majority of plants possessing CAM are either epiphytes (e.k., orchids, bromeliads) or succulent xerophytes (e.yard., cacti, cactoid Euphorbias), just CAM is also found in hemiepiphytes (eastward.g., Clusia); lithophytes (e.yard., Sedum, Sempervivum); terrestrial bromeliads; wetland plants (e.g., Isoetes, Crassula (Tillaea), Lobelia;[16] and in one halophyte, Mesembryanthemum crystallinum; one non-succulent terrestrial plant, (Dodonaea viscosa) and one mangrove associate (Sesuvium portulacastrum).
The simply copse that can practise CAM are in the genus Clusia; species of which are institute across Key America, S America and the Caribbean. In Clusia, CAM is constitute in species that inhabit hotter, drier ecological niches, whereas species living in libation montane forests tend to be Cthree [17]. In addition, some species of Clusia tin can temporarily switch their photosynthetic physiology from Ciii to CAM, a process known as facultative CAM. This allows these trees to benefit from the elevated growth rates of Cthree photosynthesis, when h2o is plentiful, and the drought tolerant nature of CAM, when the dry season occurs.
Plants which are able to switch betwixt different methods of carbon fixation include Portulacaria afra, improve known as Dwarf Jade Plant, which normally uses C3 fixation but can use CAM if it is drought-stressed,[eighteen] and Portulaca oleracea, amend known as Purslane, which usually uses C4 fixation only is also able to switch to CAM when drought-stressed.[19]
CAM has evolved convergently many times.[20] Information technology occurs in xvi,000 species (virtually 7% of plants), belonging to over 300 genera and around 40 families, but this is thought to be a considerable underestimate.[21] The great majority of plants using CAM are angiosperms (flowering plants) just it is plant in ferns, Gnetopsida and in quillworts (relatives of club mosses). Interpretation of the kickoff quillwort genome in 2022 (I. taiwanensis) suggested that its utilize of CAM was some other example of convergent development.[22]
The following list summarizes the taxonomic distribution of plants with CAM:
| Sectionalisation | Class/Angiosperm group | Order | Family | Plant Type | Clade involved |
|---|---|---|---|---|---|
| Lycopodiophyta | Isoetopsida | Isoetales | Isoetaceae | hydrophyte | Isoetes [23] (the sole genus of class Isoetopsida) - I. howellii (seasonally submerged), I. macrospora, I. bolanderi, I. engelmannii, I. lacustris, I. sinensis, I. storkii, I. kirkii, I. taiwanensis. |
| Pteridophyta | Polypodiopsida | Polypodiales | Polypodiaceae | epiphyte, lithophyte | CAM is recorded from Microsorum, Platycerium and Polypodium,[24] Pyrrosia and Drymoglossum [25] and Microgramma |
| Pteridopsida | Polypodiales | Pteridaceae[26] | epiphyte | Vittaria [27] Anetium citrifolium [28] | |
| Cycadophyta | Cycadopsida | Cycadales | Zamiaceae | Dioon edule [29] | |
| Gnetophyta | Gnetopsida | Welwitschiales | Welwitschiaceae | xerophyte | Welwitschia mirabilis [thirty] (the sole species of the order Welwitschiales) |
| Magnoliophyta | magnoliids | Magnoliales | Piperaceae | epiphyte | Peperomia camptotricha [31] |
| eudicots | Caryophyllales | Aizoaceae | xerophyte | widespread in the family; Mesembryanthemum crystallinum is a rare example of an halophyte that displays CAM[32] | |
| Cactaceae | xerophyte | Almost all cacti have obligate Crassulacean Acrid Metabolism in their stems; the few cacti with leaves may have C3 Metabolism in those leaves;[33] seedlings have Ciii Metabolism.[34] | |||
| Portulacaceae | xerophyte | recorded in approximately half of the genera (note: Portulacaceae is paraphyletic with respect to Cactaceae and Didiereaceae)[35] | |||
| Didiereaceae | xerophyte | ||||
| Saxifragales | Crassulaceae | hydrophyte, xerophyte, lithophyte | Crassulacean acid metabolism is widespread among the (eponymous) Crassulaceae. | ||
| eudicots (rosids) | Vitales | Vitaceae[36] | Cissus,[37] Cyphostemma | ||
| Malpighiales | Clusiaceae | hemiepiphyte | Clusia [37] [38] | ||
| Euphorbiaceae[36] | CAM is constitute is some species of Euphorbia [37] [39] including some formerly placed in the sunk genera Monadenium,[37] Pedilanthus [39] and Synadenium. C4 photosynthesis is likewise found in Euphorbia (subgenus Chamaesyce). | ||||
| Passifloraceae[26] | xerophyte | Adenia [twoscore] | |||
| Geraniales | Geraniaceae | CAM is found in some succulent species of Pelargonium,[41] and is also reported from Geranium pratense [42] | |||
| Cucurbitales | Cucurbitaceae | Xerosicyos danguyi,[43] Dendrosicyos socotrana,[44] Momordica [45] | |||
| Celastrales | Celastraceae[46] | ||||
| Oxalidales | Oxalidaceae[47] | Oxalis carnosa var. hirta [47] | |||
| Brassicales | Moringaceae | Moringa [48] | |||
| Salvadoraceae[47] | CAM is found in Salvadora persica.[47] Salvadoraceae were previously placed in lodge Celastrales, only are now placed in Brassicales. | ||||
| Sapindales | Sapindaceae | Dodonaea viscosa | |||
| Fabales | Fabaceae[47] | CAM is found in Prosopis juliflora (listed under the family Salvadoraceae in Sayed'south (2001) table,[47]) merely is currently in the family Fabaceae (Leguminosae) according to The Establish Listing[49]). | |||
| Zygophyllaceae | Zygophyllum [48] | ||||
| eudicots (asterids) | Ericales | Ebenaceae | |||
| Solanales | Convolvulaceae | Ipomoea [ citation needed ] (Some species of Ipomoea are C3[37] [50] - a citation is needed here.) | |||
| Gentianales | Rubiaceae | epiphyte | Hydnophytum and Myrmecodia | ||
| Apocynaceae | CAM is constitute in subfamily Asclepidioideae (Hoya,[37] Dischidia, Ceropegia, Stapelia,[39] Caralluma negevensis, Frerea indica,[51] Adenium, Huernia), and too in Carissa [52] and Acokanthera [53] | ||||
| Lamiales | Gesneriaceae | epiphyte | CAM was found Codonanthe crassifolia, but not in iii other genera[54] | ||
| Lamiaceae | Plectranthus marrubioides, Coleus [55] | ||||
| Plantaginaceae | hydrophyte | Littorella uniflora [23] | |||
| Apiales | Apiaceae | hydrophyte | Lilaeopsis lacustris | ||
| Asterales | Asteraceae[36] | some species of Senecio [56] | |||
| Magnoliophyta | monocots | Alismatales | Hydrocharitaceae | hydrophyte | Hydrilla,[36] Vallisneria |
| Alismataceae | hydrophyte | Sagittaria | |||
| Araceae | Zamioculcas zamiifolia is the simply CAM plant in Araceae, and the simply not-aquatic CAM institute in Alismatales[57] | ||||
| Poales | Bromeliaceae | epiphyte | Bromelioideae (91%), Puya (24%), Dyckia and related genera (all), Hechtia (all), Tillandsia (many)[58] | ||
| Cyperaceae | hydrophyte | Scirpus,[36] Eleocharis | |||
| Asparagales | Orchidaceae | epiphyte | Orchidaceae has more than CAM species than any other family (CAM Orchids) | ||
| Agavaceae[38] | xerophyte | Agave,[37] Hesperaloe, Yucca and Polianthes [40] | |||
| Asphodelaceae[36] | xerophyte | Aloe,[37] Gasteria,[37] and Haworthia | |||
| Ruscaceae[36] | Sansevieria [37] [47] (This genus is listed nether the family Dracaenaceae in Sayed's (2001) table, but currently in the family Asparagaceae according to The Plant List), Dracaena [59] | ||||
| Commelinales | Commelinaceae | Callisia,[37] Tradescantia, Tripogandra |
Meet also [edit]
- C2 photosynthesis
- C3 carbon fixation
- C4 carbon fixation
- RuBisCO
References [edit]
- ^ C. Michael Hogan. 2011. Respiration. Encyclopedia of Earth. Eds. Marking McGinley & C.J.cleveland. National council for Science and the Environment. Washington DC
- ^ T. de Saussure (1804). Recherches chimiques sur la végétation. Paris: Nyon.
- ^ Bonner, Walter; Bonner, James (1948). "The Role of Carbon Dioxide in Acid Formation by Succulent Plants". American Journal of Botany. 35 (2): 113. doi:10.2307/2437894. JSTOR 2437894.
- ^ Ranson S. L.; Thomas M (1960). "Crassulacean acrid metabolism" (PDF). Annual Review of Plant Physiology. 11 (i): 81–110. doi:10.1146/annurev.pp.11.060160.000501. hdl:10150/552219.
- ^ a b c Herrera, A. (2008), "Crassulacean acrid metabolism and fitness nether water deficit stress: if not for carbon gain, what is facultative CAM good for?", Annals of Botany, 103 (4): 645–653, doi:x.1093/aob/mcn145, PMC2707347, PMID 18708641
- ^ The Encyclopedia of Fruit & Nuts. CABI. 2008. p. 218.
- ^ Forseth, Irwin (2010). "The Ecology of Photosynthetic Pathways". Knowledge Projection. Nature Teaching. Retrieved 2021-03-06 .
In this pathway, stomata open at night, which allows CO2 to diffuse into the leaf to be combined with PEP and form malate. This acid is and then stored in large key vacuoles until daytime.
- ^ Ting, I P (1985). "Crassulacean Acrid Metabolism" (PDF). Almanac Review of Plant Physiology. 36 (1): 595–622. doi:10.1146/annurev.pp.36.060185.003115. hdl:10150/552219.
- ^ Raven JA, Edwards D (2001). "Roots: evolutionary origins and biogeochemical significance". Journal of Experimental Phytology. 52 (90001): 381–401. doi:10.1093/jexbot/52.suppl_1.381. PMID 11326045.
- ^ Lüttge, Ulrich (2004-06-01). "Ecophysiology of Crassulacean Acid Metabolism (CAM)". Annals of Botany. 93 (6): 629–652. doi:10.1093/aob/mch087. ISSN 0305-7364. PMC4242292. PMID 15150072.
- ^ Stanley D. SmithRussell K. MonsonJay Eastward. Anderson (1997). "CAM Succulents". Physiological Ecology of North American Desert Plants. Adaptations of Desert Organisms. pp. 125–140. doi:10.1007/978-three-642-59212-6_6. ISBN978-3-642-63900-v.
- ^ Raven, P & Evert, R & Eichhorn, Southward, 2005, "Biological science of Plants" (7th edition), p. 135 (Figure 7-26), W.H. Freeman and Company Publishers ISBN 0-7167-1007-two
- ^ a b Keeley, J (1998). "CAM Photosynthesis in Submerged Aquatic Plants". Botanical Review. 64 (2): 121–175. doi:10.1007/bf02856581. S2CID 5025861.
- ^ a b Keeley, J; Busch, G (1984). "Carbon Absorption Characteristics of the Aquatic CAM Plant, Isoetes howellii". Found Physiology. 75 (ii): 525–530. doi:10.1104/pp.76.2.525. PMC1064320. PMID 16663874.
- ^ Klavsen, S; Madsen, T (2012). "Seasonal variation in crassulacean acid metabolism past the aquatic isoetid Littorella uniflora". Photosynthesis Research. 112 (three): 163–173. doi:ten.1007/s11120-012-9759-0. PMID 22766959. S2CID 17160398.
- ^ Keddy, P.A. (2010). Wetland Environmental: Principles and Conservation. Cambridge University Press, Cambridge, Uk. p. 26.
- ^ Leverett, A.; Hurtado Castaño, N.; Ferguson, One thousand.; Winter, 1000.; Borland, A. M. (2021). "Crassulacean acid metabolism (CAM) supersedes the turgor loss point (TLP) as an of import adaptation across a precipitation gradient, in the genus Clusia". Functional Plant Biology. 48 (7): 703–716. doi:ten.1071/FP20268.
- ^ Guralnick, L. J.; Ting, I. P. (1987). "Physiological Changes in Portulacaria afra (L.) Jacq. during a Summertime Drought and Rewatering". Plant Physiology. 85 (two): 481–six. doi:10.1104/pp.85.2.481. PMC1054282. PMID 16665724.
- ^ Koch, Grand. E.; Kennedy, R. A. (1982). "Crassulacean Acid Metabolism in the Succulent C4 Dicot, Portulaca oleracea 50 Nether Natural Environmental Conditions". Found Physiology. 69 (4): 757–61. doi:ten.1104/pp.69.four.757. PMC426300. PMID 16662291.
- ^ Keeley, Jon Eastward.; Rundel, Philip Westward. (2003). "Development of CAM and C4 Carbon-Concentrating Mechanisms" (PDF). International Journal of Plant Sciences. 164 (S3): S55. doi:ten.1086/374192. S2CID 85186850.
- ^ Dodd, A. N.; Borland, A. M.; Haslam, R. P.; Griffiths, H.; Maxwell, K. (2002). "Crassulacean acid metabolism: plastic, fantastic". Journal of Experimental Botany. 53 (369): 569–580. doi:10.1093/jexbot/53.369.569. PMID 11886877.
- ^ Wickell, David; Kuo, Li-Yaung; Yang, Hsiao-Pei; others, and 11 (2021). "Underwater CAM photosynthesis elucidated by Isoetes genome". Nature Communications. 12: 6348. doi:10.1038/s41467-021-26644-7. Retrieved 29 December 2021.
- ^ a b Boston, H (1983). "Testify of crussulacean acid metabolism in ii Northward American isoetids". Aquatic Botany. 15 (4): 381–386. doi:10.1016/0304-3770(83)90006-two.
- ^ Holtum, Joseph A.M.; Winter, Klaus (1999). "Degrees of crassulacean acrid metabolism in tropical epiphytic and lithophytic ferns". Australian Periodical of Found Physiology. 26 (8): 749. doi:10.1071/PP99001.
- ^ Wong, S.C.; Hew, C.S (1976). "Diffusive Resistance, Titratable Acidity, and CO2 Fixation in 2 Tropical Epiphytic Ferns". American Fern Periodical. 66 (four): 121–124. doi:10.2307/1546463. JSTOR 1546463.
- ^ a b Crassulacean Acid Metabolism Archived 2007-06-09 at the Wayback Machine
- ^ abstract to Carter & Martin, The occurrence of Crassulacean acid metabolism among ephiphytes in a high-rainfall region of Costa Rica, Selbyana fifteen(2): 104-106 (1994)
- ^ Martin, Shannon L.; Davis, Ryan; Protti, Piero; Lin, Teng-Chiu; Lin, Shin-Hwei; Martin, Craig Eastward. (2005). "The Occurrence of Crassulacean Acid Metabolism in Epiphytic Ferns, with an Emphasis on the Vittariaceae". International Journal of Plant Sciences. 166 (4): 623–630. doi:10.1086/430334. S2CID 67829900.
- ^ Vovides, Andrew P.; Etherington, John R.; Dresser, P. Quentin; Groenhof, Andrew; Iglesias, Carlos; Ramirez, Jonathan Flores (2002). "CAM-cycling in the cycad Dioon edule Lindl. in its natural tropical deciduous forest habitat in central Veracruz, Mexico". Botanical Periodical of the Linnean Order. 138 (ii): 155–162. doi:10.1046/j.1095-8339.2002.138002155.ten.
- ^ Schulze, E. D.; Ziegler, H.; Stichler, W. (1976). "Environmental command of crassulacean acid metabolism in Welwitschia mirabilis Claw. Fil. in its range of natural distribution in the Namib desert". Oecologia. 24 (4): 323–334. Bibcode:1976Oecol..24..323S. doi:x.1007/BF00381138. PMID 28309109. S2CID 11439386.
- ^ Sipes, DL; Ting, IP (1985). "Crassulacean Acid Metabolism and Crassulacean Acid Metabolism Modifications in Peperomia camptotricha". Plant Physiology. 77 (one): 59–63. doi:x.1104/pp.77.1.59. PMC1064456. PMID 16664028.
- ^ Chu, C; Dai, Z; Ku, MS; Edwards, GE (1990). "Consecration of Crassulacean Acid Metabolism in the Facultative Halophyte Mesembryanthemum crystallinum by Abscisic Acid". Institute Physiology. 93 (iii): 1253–1260. doi:10.1104/pp.93.3.1253. PMC1062660. PMID 16667587.
- ^ Nobel, Park S.; Hartsock, Terry Fifty. (1986). "Foliage and Stalk COtwo Uptake in the Three Subfamilies of the Cactaceae". Establish Physiology. 80 (iv): 913–917. doi:10.1104/pp.80.four.913. PMC1075229. PMID 16664741.
- ^ Wintertime, One thousand.; Garcia, Yard.; Holtum, J. A. 1000. (2011). "Drought-stress-induced upwardly-regulation of CAM in seedlings of a tropical cactus, Opuntia elatior, operating predominantly in the C3 way". Journal of Experimental Botany. 62 (11): 4037–4042. doi:x.1093/jxb/err106. ISSN 0022-0957. PMC3134358. PMID 21504876.
- ^ Guralnick, Lonnie J.; Jackson, Michael D. (2001). "The Occurrence and Phylogenetics of Crassulacean Acrid Metabolism in the Portulacaceae". International Journal of Plant Sciences. 162 (two): 257–262. doi:10.1086/319569. S2CID 84007032.
- ^ a b c d e f g Cockburn, Due west. (1985). "TANSLEY REVIEW No ane.. VARIATION IN PHOTOSYNTHETIC ACID METABOLISM IN VASCULAR PLANTS: CAM AND RELATED PHENOMENA". New Phytologist. 101 (1): 3–24. doi:10.1111/j.1469-8137.1985.tb02815.x. PMID 33873823.
- ^ a b c d e f thousand h i j 1000 Nelson, Elizabeth A.; Sage, Tammy L.; Sage, Rowan F. (2005). "Functional leaf anatomy of plants with crassulacean acid metabolism". Functional Plant Biology. 32 (5): 409–419. doi:10.1071/FP04195. PMID 32689143.
- ^ a b Lüttge, U (2004). "Ecophysiology of Crassulacean Acid Metabolism (CAM)". Annals of Botany. 93 (vi): 629–52. doi:ten.1093/aob/mch087. PMC4242292. PMID 15150072.
- ^ a b c Bender, MM; Rouhani, I.; Vines, H. One thousand.; Black, C. C. (1973). "C/C Ratio Changes in Crassulacean Acid Metabolism Plants". Plant Physiology. 52 (5): 427–430. doi:ten.1104/pp.52.5.427. PMC366516. PMID 16658576.
- ^ a b Szarek, S.R. (1979). "The occurrence of Crassulacean Acid Metabolism a supplementary list during 1976 to 1979". Photosynthetica. 13 (4): 467–473.
- ^ Jones, Cardon & Czaja (2003). "A phylogenetic view of low-level CAM in Pelargonium (Geraniaceae)". American Periodical of Botany. ninety (ane): 135–142. doi:10.3732/ajb.90.1.135. PMID 21659089.
- ^ M. Kluge, I. P. Ting (2012). Crassulacean Acid Metabolism: Analysis of an Ecological Adaptation Volumen 30 de Ecological Studies. Springer Science & Concern Media. p. 24. ISBN9783642670381.
- ^ Bastide, Sipes, Hann & Ting (1993). "Effect of Severe Water Stress on Aspects of Crassulacean Acid Metabolism in Xerosicyos". Plant Physiol. 103 (4): 1089–1096. doi:10.1104/pp.103.4.1089. PMC159093. PMID 12232003.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gibson, Arthur C. (2012). Structure-Function Relations of Warm Desert Plants Adaptations of Desert Organisms. Springer Science & Business Media. p. 118. ISBN9783642609794.
- ^ "Momordica charantia (bitter melon): 111016801". Kyoto Encyclopedia of Genes and Genomes.
- ^ Bareja, Ben G. (2013). "Plant Types: Three. CAM Plants, Examples and Plant Families". Cropsreview.
- ^ a b c d e f yard Sayed, O.H. (2001). "Crassulacean Acid Metabolism 1975–2000, a Check List". Photosynthetica. 39 (3): 339–352. doi:ten.1023/A:1020292623960. S2CID 1434170.
- ^ a b R. MATTHEW OGBURN, ERIKA J. EDWARDS (2010). "The Ecological Water-Use Strategies of Delicious Plants" (PDF). Brown University.
- ^ "Prosopis juliflora". The Plant Listing . Retrieved 2015-09-11 .
- ^ Craig Due east. Martin; Anne E. Lubbers; James A. Teeri (1982). "Variability in Crassulacean Acid Metabolism A Survey of Due north Carolina Delicious Species". Botanical Gazette. 143 (iv): 491–497. doi:x.1086/337326. hdl:1808/9891. JSTOR 2474765. S2CID 54906851.
- ^ Lange, Otto L.; Zuber, Margit (1977). "Frerea indica, a stem succulent CAM plant with deciduous C3 leaves". Oecologia. 31 (1): 67–72. Bibcode:1977Oecol..31...67L. doi:10.1007/BF00348709. PMID 28309150. S2CID 23514785.
- ^ Madhusudana Rao, P.M Swamy, Five.Due south.R Das (1979). "Some Characteristics of Crassulacean Acid Metabolism in Five Nonsucculent Scrub Species Under Natural Semiarid Conditions". Zeitschrift für Pflanzenphysiologie. 94 (three): 201–210. doi:x.1016/S0044-328X(79)80159-ii.
{{cite journal}}: CS1 maint: multiple names: authors listing (link) - ^ Houérou, Henry North. (2008). Bioclimatology and Biogeography of Africa Earth and Environmental Scientific discipline. Springer Science & Business concern Media. p. 52. ISBN9783540851929.
- ^ Guralnick; Ting, Irwin P; Lord, Elizabeth M; et al. (1986). "Crassulacean Acid Metabolism in the Gesneriaceae". American Journal of Botany. 73 (3): 336–345. doi:10.2307/2444076. JSTOR 2444076.
- ^ One thousand. V. Ramana and K. V. Chaitanya (December 2014). "Variations in 13C Rates and Crassulacean Acid Metabolism of Six Coleus species" (PDF). journalrepository.org.
- ^ Fioretti & Alfani; Alfani, A (1988). "Anatomy of Succulence and CAM in 15 Species of Senecio". Botanical Gazette. 149 (2): 142–152. doi:10.1086/337701. JSTOR 2995362. S2CID 84302532.
- ^ Holtum, Winter, Weeks and Sexton (2007). "Crassulacean acid metabolism in the ZZ plant, Zamioculcas zamiifolia (Araceae)". American Periodical of Phytology. 94 (x): 1670–1676. doi:10.3732/ajb.94.x.1670. PMID 21636363.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Crayn, D. Grand.; Winter, Thou; Smith, JA (2004). "Multiple origins of crassulacean acid metabolism and the epiphytic habit in the Neotropical family Bromeliaceae". Proceedings of the National University of Sciences. 101 (x): 3703–8. Bibcode:2004PNAS..101.3703C. doi:10.1073/pnas.0400366101. PMC373526. PMID 14982989.
- ^ Silvera, Katia & Neubig, Kurt & Whitten, William & Williams, Norris & Winter, Klaus & C. Cushman, John. "Evolution along the crassulacean acrid metabolism continuum". Research gate.
{{cite web}}: CS1 maint: multiple names: authors list (link)
External links [edit]
| | Wikimedia Commons has media related to CAM cycle. |
- Khan Academy, video lecture
Source: https://en.wikipedia.org/wiki/Crassulacean_acid_metabolism
0 Response to "Cam Plants Keep Stomata Closed in Daytime Thus Reducing Loss of Water. They Can Do This Because They"
Post a Comment